ISO 13485, audit-ready by default.
CAPA, NCR, Change Control, Audits, Complaints, Risk Register, Design Reviews, Test Management, Training. Signed change control. Immutable audit log. Traceability from requirement to release — without a quality engineer to babysit it.
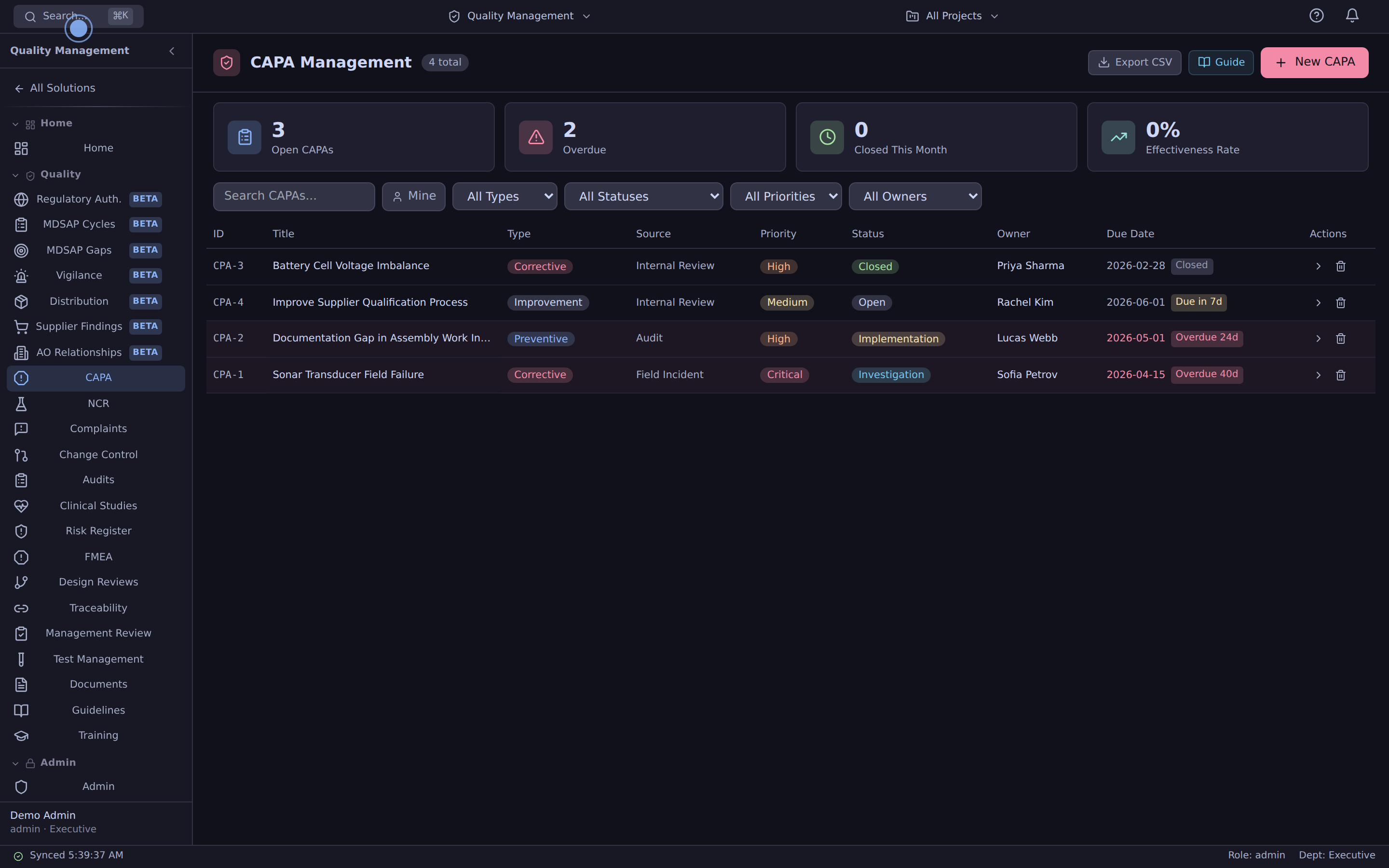
CAPA, NCR, change control — one database, not seven.
Most QMS tools are document repositories with a workflow bolted on. Azora's QMS lives in the same database as your tasks, units, and vendors — every CAPA links to the bug it traces, every audit finding links to the document it concerns.
CAPA management
Corrective & preventive actions with effectiveness checks. Auto-link to source NCRs, complaints, audit findings.
NCR + Change Control
Nonconformance reports with disposition workflow. Change control with electronic signatures (21 CFR Part 11).
Document control
Versioned documents with approval workflow. Read-receipt tracking. Periodic review cadence.
Audit management
Internal + external audits. Finding tracking. Mock external audit prep for MDSAP cycles.
Risk register (ISO 14971)
FMEA-style risk analysis. Severity × probability × detectability scoring. Linked to design controls.
Traceability matrix
Every requirement → spec → test → release. Click any node, the whole chain lights up.
"Our auditor noted that our traceability matrix was the most complete he'd seen in a Class II company under 50 people. We didn't build it — Azora generates it from links we'd be making anyway."
Nadia · Regulatory Lead at Azora Optical Solutions — the team that built Azora1 DB
CAPAs, NCRs, audits — one query
100%
Audit findings traceable to source
0
External tools required for audit pack
See your QMS without the spreadsheets.
Sign up, import your existing CAPA log via CSV, and you'll have a connected quality system within an afternoon.